 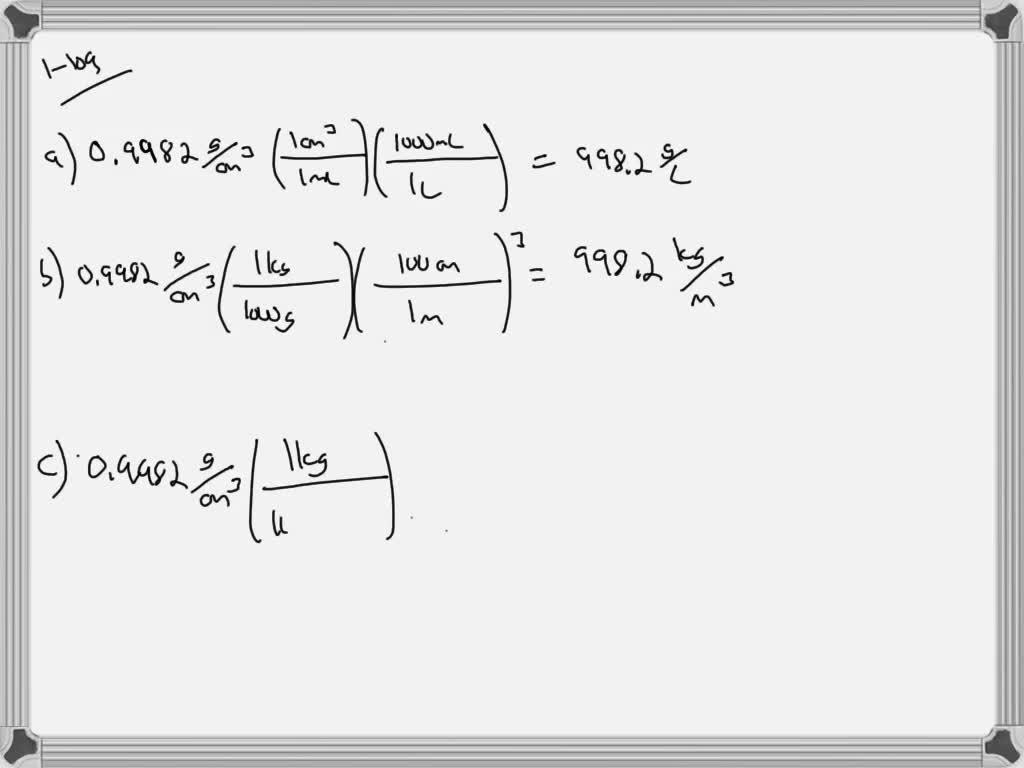
Some materials like Silicon, Germanium, Bismuth, and water are less dense in solid form than in liquid form. 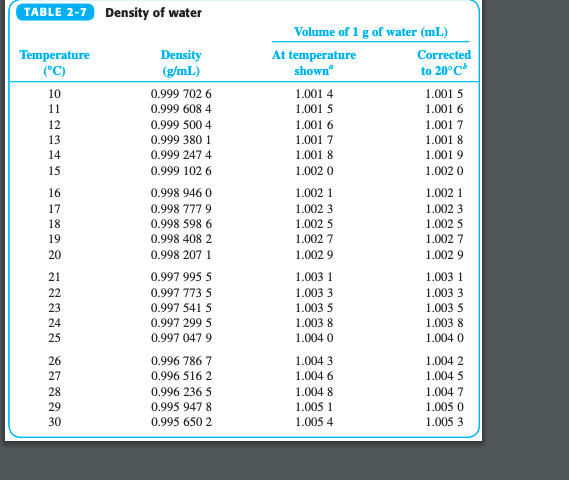
Therefore, the ρ solid > ρ liquid > ρ gas The atoms or molecules of solids are more closers together than the atoms or molecules of the liquid. Generally, when a solid mass of a substance is put in a liquid of the same substance, it sinks. Since the ρ water of ice is less than the ρ water of water, therefore ice floats on water. Less dense matter floats on the dense liquid. The density of water (g/ml) = 8kg/(8 x10 -3 m 3 ) The mass (m) of 8 liters of water is 8 kg. Γ water = ρ water x g = 1000 kg/m 3 x 9.807 m/sec 2 = 9.807 kN/m 3 ( 1 N = kg⋅m/s 2) Density Of Water in g/ml-Example Problem Specific weight (γ) = (ρ x volume x g)/volume The specific weight of water on Earth at 4☌ = 9.807 kN/m³ = 62.43 lbf/ft³. The specific weight of water refers to the weight per unit volume of a material. The density of fresh water on Earth at 4☌ is normally taken as 1000 kg/m 3. Common units of ρwater are grams per milliliter (1 g/ml), grams per cubic centimeter ( g/cm3), and pounds per cubic foot (lb/ft 3). The density of water ( ρ water )is around 1g/ml, but this value changes with temperature or impurities present in water. This is due to the large quantity of matter in iron than in wood.ĭensity (ρ) formula = mass (m) of substance ÷ volume (v) of substance The density of water in g/ml We will find that iron is heavier than wood. If we take equal volumes of wood and iron. It is the ratio of mass to volume of a substance. How much does a gallon of water weigh at its highest density?ĭensity is the quantity of matter per unit volume.The density of water in g/ml (effect of temperature). 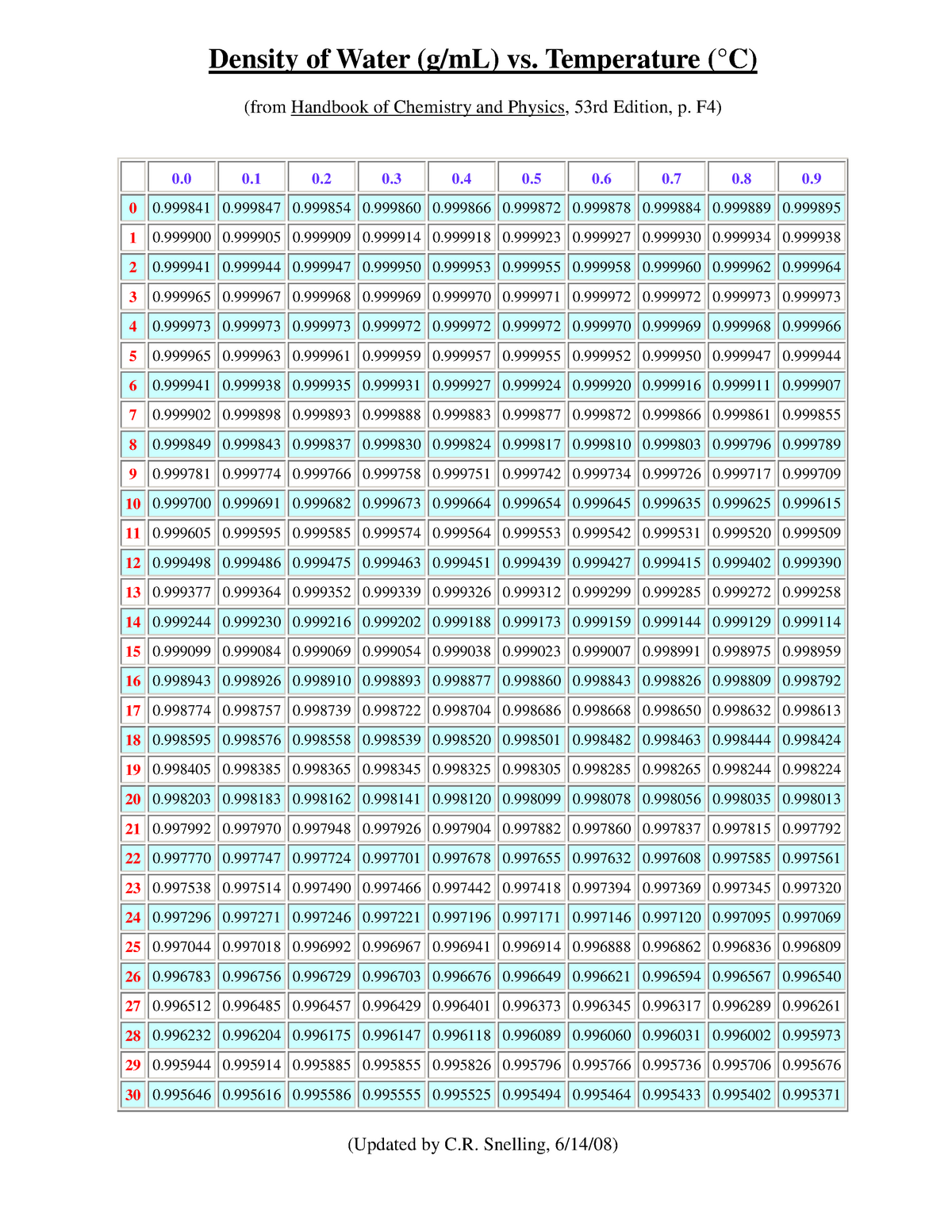
Density Of Water in g/ml-Example Problem.Calculating the specific weight of water.Keiko wants to pour a total of milliliters of a liquid. How many more milliliters must Keiko pour? If the water is at standard temperature pressure, then weight in grams = volume in ml Subtract. Calculate the density of liquid x And Volume of density bottle? Should be 30 degrees celsius due to the equation Q=MC(delta)T.Īn empty density bottle weighs 30g.when completely filled with water,it weighs 75g and when completely filled with liquid x weighs 65g. Pour a liter of water at 40 celsius into a liter of water at 20 celsius and the final temperature of the two becomes about _C? 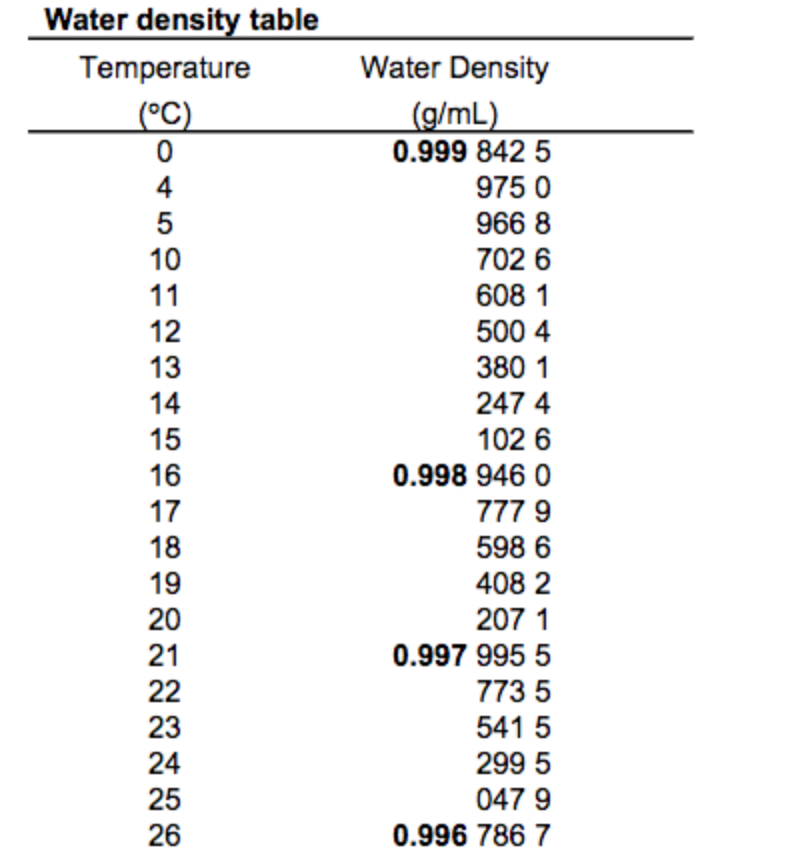
Using these you can calculate and compare. You need a calculator, a triple beam balance and a cylinder. Which Set Of Equipment Would Be Most Useful To Determine The Density Of A Liquid? M=3.75/1.17=3.2 Liters, so the volume of the liquid is 3.2 Liters. If A Liquid Has A Density Of 1.17 G/cm3 How Many Liters Of The Liquid Have A Mass Of 3.75 Kg? The density of a substance is equal to the ratio of mass of substance to the volume. What Is The Volume Of 28.1 G Of Liquid Bromine? Why does gas have a lower density than a liquid.īromine Is A Red Liquid At 25✬. Why Does Gas Have A Lower Density Than A Liquid? As I looked on the Internet it seemed to be the same. The equation for density is Density=Mass/Volume. What Are The Steps To Finding The Density Of A Solid? Of A Liquid? 92 G/cm3 What Is The Volume Of A 5000 Gram Bag Of Ice? The common way to define the volume or density of a powder is through the packing volume which refers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
